IR spectroscopy
- 1. Infrared Spectroscopy By: Bijaya Kumar Uprety
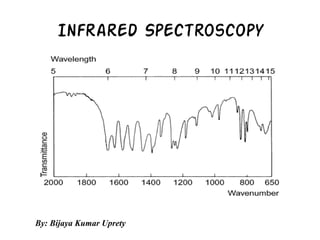
- 2. Introduction •Thetwoatomsjoinedtogetherbyachemicalbond(maybesingle,doubleortriplebond),macroscopicallycanbecomposedastwoballsjoinedbyaspring. •Theapplicationofaforcelike(i)stretchingofoneorboththeballs(atoms)awayfromeachotherorclosertoeachother(ii)bendingofoneoftheatomseitherverticallyorhorizontallyandthenreleaseoftheforceresultsinthevibrationsonthetwoballs(atoms). •Thesevibrationsdependonthestrengthofthespringandalsothemode(stretchingorbending)inwhichtheforceisbeingapplied. •Similarly,atordinarytemperatures,organicmoleculesareinaconstantstateofvibrations,eachbondhavingitscharacteristicstretchingandbendingfrequencies. •Wheninfraredlightradiationsbetween4000-400cm-1(theregionmostconcernedtoanorganicchemist)arepassedthroughasampleofanorganiccompound, someoftheseradiationsareabsorbedbythesampleandareconvertedintoenergyofmolecularvibrations.Theotherradiationswhichdonotinteractwiththesamplearetransmittedthroughthesamplewithoutbeingabsorbed.Theplotof% transmittanceagainstfrequencyiscalledtheinfraredspectrumofthesampleorcompound.
- 3. Continue… •Thisstudyofvibrationsofbondsbetweendifferentatomsandvariedmultiplicitieswhichdependingontheelectronegativity,massesoftheatomandtheirgeometryvibrateatdifferentbutspecifiedfrequencies;iscalledinfraredspectroscopy. •Thepresenceofsuchcharacteristicvibrationalbandsinaninfraredspectrumindicatesthepresenceofthesebondsinthesampleunderinvestigation. Principle: IRspectroscopyisbasedupontheprinciplethat,whenacompoundisexposedtoIRradiations,itselectivelyabsorbstheradiationsresultinginvibrationofthemoleculesofthecompound.Itresultsincloselypackedabsorptionbandswhicharecharacteristictothefunctionalgroupsandbondspresentinthecompound.ThusanIRspectrumofcompoundisconsideredasthefingerprintforitsidentification.
- 4. •Theinfraredportionoftheelectromagneticspectrumisusuallydividedintothreeregions;thenear-,mid- andfar-infrared,namedfortheirrelationtothevisiblespectrum. •Thehigher-energynear-IR,approximately14000– 4000cm−1(0.8–2.5μmwavelength)canexciteovertoneorharmonicvibrations. •Themid-infrared,approximately4000–400cm−1(2.5– 25μm)maybeusedtostudythefundamentalvibrationsandassociatedrotational- vibrationalstructure. •Thefar-infrared,approximately400–10cm−1(25– 1000μm),lyingadjacenttothemicrowaveregion,haslowenergyandmaybeusedforrotationalspectroscopy.
- 5. Hooke’s law and Absorption of radiations • •Therefore,inprinciple,eachabsorptionofradiationintheinfraredregionisquantizedandshouldappearassharpline.However,eachvibrationaltransitionwithinthemoleculeisassociatedwithnumberofrotationalenergychangesandthusappearsascombinationofvibrational-rotationalbands. •Theanalogyofachemicalbondwithtwoatomslinkedthroughaspringcanbeusedtorationalizeseveralfeaturesoftheinfraredspectroscopy. •TheapproximationtovibrationfrequencyofabondcanbemadebytheapplicationofHooke’slaw.InHooke’slaw,twoatomsandtheirconnectingbondaretreatedasasimpleharmonicoscillatorcomposedoftwomassesjoinedbyaspringandfrequencyofvibrationisstatedas
- 8. IR Frequency Range •Infraredradiationspansasectionoftheelectromagneticspectrumhavingwavenumbersfromroughly13,000to10cm–1,orwavelengthsfrom0.78to1000μm.Itisboundbytheredendofthevisibleregionathighfrequenciesandthemicrowaveregionatlowfrequencies. •IRabsorptionpositionsaregenerallypresentedaseitherwavenumbersorwavelengths(l). •Wavenumberdefinesthenumberofwavesperunitlength.Thus,wavenumbersaredirectlyproportionaltofrequency,aswellastheenergyoftheIRabsorption. Thewavenumberunit(cm–1,reciprocalcentimeter)ismorecommonlyusedinmodernIRinstrumentsthatarelinearinthecm–1scale. •Inthecontrast,wavelengthsareinverselyproportionaltofrequenciesandtheirassociatedenergy. •Wavenumbersandwavelengthscanbeinterconvertedusingthefollowingequation:
- 9. •ThefarIRrequires[400–10cm−1(25–1000μm)]theuseofspecializedopticalmaterialsandsources.Itisusedforanalysisoforganic,inorganic, andorganometalliccompoundsinvolvingheavyatoms(massnumberover19).Itprovidesusefulinformationtostructuralstudiessuchasconformationandlatticedynamicsofsamples. •NearIR[14000–4000cm−1(0.8–2.5μmwavelength)]needsminimalornosamplepreparation.Itoffershigh-speedquantitativeanalysiswithoutconsumptionordestructionofthesample.ItsinstrumentscanoftenbecombinedwithUV-visiblespectrometerandcoupledwithfiberopticdevicesforremoteanalysis.NearIRspectroscopyhasgainedincreasedinterest,especiallyinprocesscontrolapplications. •However,themostfrequentlyusedregionismidIRregionwhichliesbetween4000and400cm–1.
- 10. Region of IR Spectrum •A molecule absorbs IR radiations of various wavelengths depending upon the nature of groups or bonds present in it and gives characteristic absorption bands. •The presence of characteristic absorption band indicates the presence of a particular functional group. •There are two general regions in the infrared spectrum, namely : (a)Groupfrequencyregion/FunctionalGroupRegion:havingawavenumberfrom4000-1500cm–1.Here,thestretchingandbendingvibrationalbandsassociatedwithspecificstructuralorfunctionalgroupsareobservedfrequently. (b)Fingerprintregion:havingawavenumberfrom1500-400cm–1.Here,thevibrationalmodesdependsolelyandstronglyontherestofthemolecule.Example:TheC—Cstretchingfrequencydependslargelyonwhatelseisbondedtothecarbonatoms.Itisinterestingtoobserveherethatthisparticularregionofthespectrumisdenselypopulatedwithbands.Asweknowthatnotwo‘fingerprints’couldbeidenticalinhumanbeings,exactlyinasimilarmannernotwocompoundsmayhavethesame‘fingerprintregion’.Thus,eachandeverymoleculeessentiallygivesrisetoauniquespectrumwhichoffersacharacteristicfeatureofthesame.
- 11. The primary regions of the IR spectrum
- 12. Criteria for Absorption of IR radiation (i)DipoleMoment: •Weknowthatatordinarytemperature,moleculesareinconstantstateofvibrations. Thechangeindipolemomentduringvibrationofthemoleculeproducesastationaryalternatingelectricfield. •Whenthefrequencyofincidentelectromagneticradiationsisequaltothealternatingelectricfieldproducedbychangesinthedipolemoment,theradiationisabsorbedandvibrationallevelsofthemoleculeareexcited. •Onceinthevibrationallyexcitedstate,themoleculescanloosetheextraenergybyrotational,collisionortranslationalprocessesetc.andcomebacktogroundstate. •Therefore,onlythosevibrationswhichresultinarhythmicalchangeinthedipolemomentofthemoleculeabsorbinfraredradiationsandaredefinedasIRactive. •TheotherswhichdonotundergochangeindipolemomentofthemoleculeareIRinactivee.g.thestretchingofasymmetricallysubstitutedbond,vizC≡Cinacetyleneandsymmetricalstretchingincarbondioxide,alinearmolecule,producenochangeinthedipolemomentofthesystemandthesevibrationscannotinteractwithinfraredlightandareIRinactive. •Ingeneral,thefunctionalgroupsthathaveastrongdipolegiverisetostrongabsorptionbandsintheIR.
- 14. Modes of Vibration •Allmoleculesarecontinuallyvibrating.Thesevibrationareoftwotypes: A.Stretchingvibrations:Inthistypeofvibration,thedistancebetweenthetwoatomsincreasesordecreases,buttheatomsremaininthesamebondaxis.Thisisduetochangeinbondlength.Thestretchingvibrationsmaybefurthersub-dividedintotwocategories,namely: (a)SymmetricalStretching:Inthiscase,thetwoatoms(AandB)eithermovetowardsorawayfromthecentralatom,withoutchangeinbondaxisorbondangle. (b)AsymmetricalStretching:Inthisinstance,thetwoatomsAandBmovewithrespecttocentralatomsuchthatonemovesawayandtheothermovestowardsthecentralatomC. B.BendingVibration:Thesetypeofvibrationinvolvesmovementofatomswhichareattachedtoacommoncentralatom,suchthatthereischangeinbondaxisandbondangleofeachindividualatomwithoutchangeintheirbondlength.Bendingvibrationscantakeplaceeitherin-planeorout-of-plane.
- 15. (i) In-Plane Bending Vibrations These are two types : (a) Scissoring or Symmetrical Bending : In these type of vibration, in plane bending of atoms occur wherein they move back and forth. (b) Rocking : In this case, in plane bending of atoms occur wherein they swing back and forth with respect to the central atom. (ii) Out-of Plane Bending Vibrations These are also of two kinds, namely : (a) Wagging : In these type of vibrations, out of plane bending of atoms occur wherein they oscillate back and forth. (b) Twisting : In these types of vibrations, out of plane bending of atoms occur wherein they rotate around the bond which joins the central atom C of molecule.
- 16. •Eachbendingandstretchingvibrationofabondinamoleculeoccurswithacharacteristicfrequency. •Whenacompoundisbombardedwithradiationofafrequencythatexactlymatchesthefrequencyofoneofitsvibrations,themoleculewillabsorbenergy. •Determiningthewavenumbersoftheenergyabsorbedbyaparticularcompound,wecantellwhatkindsofbondsithas. •The bending (or deformation) vibrations generally require less energy and take place at longer-wavelength than the corresponding stretching vibrations. •In contrast, the stretching vibrations are observed to occur with respect to their corresponding bond strengths. Note: v(asymmetricstretching)>v(symmetricstretching)>v(bending)
- 19. Factors influencing vibrational Frequencies •There are a number of factors that influence the precise frequency of a molecular vibration, namely : (a) Vibrational coupling, (b) Hydrogen bonding, (c) Electronic Effects, and (d) Field Effects. a.Vibrational coupling: •Vibrational coupling involves interaction between two similar or different vibrating groups. Vibrational coupling depends upon various factors such as: 1.When two atoms are bonded to a common central atom, the stretching vibrations of these atoms show strong coupling. 2.Coupling between bending vibrations is seen only when the two vibrating groups are separated by a common bond. 3.Coupling is maximum when the vibrating groups have individual energies which are approximately equal in magnitudes. 4.If the vibrating groups are separated by two or more bonds, negligible or no coupling takes place.
- 20. •Example1:VibrationcouplingcanbebestexplainedbytakinganexampleoftriatomicmoleculesuchasCO2.AsCO2isalinearmolecule,thenumberofmodesofvibrationsforitisgivenby; 3n-5=3x3-5=4.ThusnumberofpeaksshouldbefourintheIRspectrum(twostretchingandtwobending) •Buthereonlytwopeaksappearthatisanti-symmetricandbendingpeaks. •Anti-symmetricpeaksappearat2350cm-1. •Heresymmetricpeakdoesn’tappearbecausethereisnochangeindipolemoment.ThereforeitisIRinactive. •Andinsteadoftwobendingpeakonlyonebendingpeakappearsbecauseenergyofabsorptionissame. •Example2:Anothere.g.isofAcetaldehyde.Heren=7andmoleculeisnon-linearsothenumberofpeaksaccordingtoformula3n-6shouldbe15butonlyfivepeaksappearinthespectraduetovibrationalcoupling.
- 21. (b)HydrogenBonding •HydrogenbondingisatypeofelectrostaticinteractionbetweenmoleculesthathavehydrogenatomandelectronegativeatomslikeF,N, Oetc. •Compoundscontainingprotondonorgroupssuchascarbonyl,hydroxylamine(NH2-OH),amide(CO-NH2),etccanbeinvolvedinintra-orintermolecularhydrogenbondinginthepresenceofprotonacceptors, e.g.O,N,halogens,C=C. •ThestiffnessoftheX—Hbondistherebylessened,resultinginaloweringofthestretchingfrequency,andthebandbroadensandoftenintensifies. •Conversely,thefrequencyofthebendingmodeisraisedbuttheeffectismuchlesspronounced. •Intermolecularhydrogenbondingissuppressedatelevatedtemperaturesbutitisfavouredbyahighsoluteconcentration. •Intramolecularhydrogenbondingisalsoreducedatelevatedtemperaturesbutitisunaffectedbychangesinsoluteconcentration. Theseeffectsareparticularlysignificantinthespectraofalcohols, phenols,carboxylicacidsandamines.
- 22. •GroupssuchashydroxylandamineshowscharacteristicvibrationalfrequencieswhenexposedtoIRradiations.However,thesefrequenciesarealteredwhenthecompoundcontainingthesegroupsexhibitshydrogenbonding. •E.g.Hydrogenbondinginanalcohols(-OH)isstrongerthanthatinamines(-NH2)because,oxygenofalcoholismoreelectronegativethannitrogenofamines. •Thus,vibrationalfrequencyof(-OH)groupisloweredmoreinIRspectrumofalcoholswhencomparedto(-NH2)groupofamines. •Strengthofhydrogenbondingisinverselyproportionaltothechangeinfundamentalvibrationalfrequencyofthecompoundinvolvedinbonding.i.e.ifthehydrogenbondingisstrong,then, thefundamentalvibrationalfrequencyofthegroupinvolvedinbondingisloweredandtheabsorptionbandobtainedisbroaderandmoreintenseandviceversa.
- 23. (c)ElectronicEffect: •Dependsonthepresenceofsubstituent. •Inorganicmolecules,theelectrondisplacementtakesplacebyvariousmechanismsuchasConjugationeffect,mesomericeffectandinductiveeffects. •ThesemechanisminfluencesthevibrationfrequenciesinIRspectroscopy. 1.Conjugationeffect: •ItisobservedtolowerthefrequencyofbothC=Cstr.andC=Ostr. •In II, lowering of band order in the C = C bond is observed due to conjugation effect whereby the stretching vibration frequency is decreased by 40-50 cm–1(compare I and II above).
- 24. •Inasimilarmanner,inVdelocalizationofπ-electronsbetweenC=Oandthebenzeneringenhancesthedouble-bondcharacterofthebondjoiningtheC=Otothering.ItultimatelyleadstoalowerbandorderintheC=Obondtherebydecreasingthestretchingvibrationfrequencyby20-30cm–1(compareIIIandVabove). 2.Resonanceeffect(Mesomericeffect): •Meanssinglemoleculecanberepresentedintwoormorethantwostructuresthatdifferonlyinthearrangementofelectrons. •Ifelectronreleasinggrouppresentincreasesindelocalizationofelectronitdecreasesthedoublebondcharacteristicincreaseinbondlengthdecreaseinbondstrengthsodecreasesvibrationalfrequency. •Similarly,ifelectronwithdrawinggrouppresentitincreasesvibrationalfrequency.
- 26. 3.InductiveEffect: •Theinductiveeffectssolelydependsuponthe‘intrinsic’tendencyofasubstituenttoeitherreleaseorwithdrawelectrons,itselectronegativityactingeitherthroughthemolecularchainorthroughspace.Thiseffectusuallyweakenssteadilywithincreasingdistancefromthesubstituent. •Inductiveandresonancebothtypeofeffectsareexistinginmolecule. •Finallywhichtypeofeffectisshownbyamoleculedependsonwhicheffectisprominent. e.g. i.Amides:(R-C-NH2):Containsmoreelectronreleasingmoiety(NH2) thusenhancedresonanceeffectsodecreasedvibrationalfrequency. Fig(q) ii.Acylchloride(R-CO-Cl):C=Oiselectronwithdrawingsodecreaseindelocalizationofelectronincreasedbondstrengthincreasevibrationalfrequency.Fig(r)
- 28. 4. Field Effect
- 29. Instrumentation •Theimportantfeaturesofaninfraredspectrophotometerareasfollows: (i)Infraredsources, (ii)Monochromators, (iii)Detectors,and (iv)ModeofOperation InfraredSources •Themostcommoninfraredsourcesareelectricallyheatedrodsofthefollowingtypes: (a)FusedmixturesoftheoxidesofZirconium(Zr),Yttrium(Y),Erbium(Er)etc., alsoknownas‘NernstGlower’, (b)SiliconCarbide‘Globar’,and (c)Variousceramic(clay)materials. •Itisquiteevidentthattheinfraredoutputfromallthesedifferentsourcesinvariablyvariesinintensityoveradefinitefrequencyrange,therefore,acompensatingvariableslitisusuallyprogrammedtooperateinunisonwiththescanningovertheindividualfrequencies.
- 30. Monochromators •Threetypesofsubstancesarenormallyemployedasmonochromators,namely: (i)MetalHalidePrisms:Variousmetalhalideprisms,suchas:KBr(12- 25μm),LiF(0.2-6μm)andCeBr(15-38μm)havebeenusedearlier, buttheyhavebecomemoreorlessobsolescentnowadays. (ii)NaClPrism(2-15μm):Sodiumchlorideprismareofuseforthewholeoftheregionfrom4000-650cm–1.First,itofferslowresolutionat4000-2500cm–1,andsecondly,becauseofitshygroscopicnaturetheopticshavegottobeprotectedat20°Cabovetheambienttemperature. (iii)Gratings:Ingeneral,gratingsarecommonlyemployedinthedesignoftheinstrumentsandofferbetterresolutionathigherfrequencythantheprisms.Theyoffermuchbetterresolutionatlowfrequency,viz.,typicalrulingsare240linespernmforthe4000- 1500cm–1regionand120linespernmforthe1500-650cm–1region.
- 31. Detectors •Therearethreedifferenttypesofdetectorsthatareusedintheinfraredregion: (a)Thermocouples(orThermopiles):Theunderlyingprincipleofathermocoupleisthatiftwodissimilarmetalwiresarejoinedheadtotail,thenadifferenceintemperaturebetweenheadandtailcausesacurrenttoflowinthewires.Intheinfraredspectrophotometerthiscurrentshallbedirectlyproportionaltotheintensityofradiationfallingonthethermocouple.Hence,thethermocouplesareinvariablyemployedintheinfraredregion,andtohelpinthecompleteabsorptionof‘availableenergy’the‘hot’junctionorreceiverisnormallyblackened. (b)GolayDetector:Inthisspecificinstancetheabsorptionofinfraredradiationaffordsexpansionofaninertgasinacell-chamber.Theheatproducedduetoabsorptionofradiationcausesthegastoexpandandthusdeformtheflexiblesilvereddiaphragmwhichactsasmirror.Thedeformationofdiaphragmisamplifiedbyplacingalampinthedetector.Fromthelamp,lightismadetofallonthediaphragmwhichinturnreflectsitontothephototube.Whendiaphragmisatrestnolightpassesthroughitbutwhendiaphragmflexesvaryingamountoflightreachesphototube.Thecurrentfromthephototubeisdirectlyproportionaltotheincidentradiation.
- 33. (c)Bolometers:Thesearebasedontheprinciplethatmakeuseoftheincreaseinresistanceofametalwithincreaseintemperature.Forinstance,whenthetwoplatinumfoilsareappropriatelyincorporatedintoaWheatstonebridge,andradiationisallowedtofallononeofthefoil(knownasindicatorstrip),achangeintheresistanceisobservedultimately.Thiscausesanout-of-balancecurrentthatisdirectlyproportionaltotheincidentalradiation.Justlikethethermocouples,theyareusedintheinfraredregion. TypesofIRspectrometer Theinstrumentthatdeterminestheabsorptionspectrumforacompoundiscalledaninfraredspectrometeror,moreprecisely,aspectrophotometer.Twotypesofinfraredspectrometersareincommonuseintheorganiclaboratory:dispersiveandFouriertransform(FT) instruments.Bothofthesetypesofinstrumentsprovidespectraofcompoundsinthecommonrangeof4000to400cm−1.Althoughthetwoprovidenearlyidenticalspectraforagivencompound,FTinfraredspectrometersprovidetheinfraredspectrummuchmorerapidlythanthedispersiveinstruments.
- 34. Dispersive Infrared Spectrometers •Figure2.3aschematicallyillustratesthecomponentsofasimpledispersiveinfraredspectrometer.Theinstrumentproducesabeamofinfraredradiationfromahotwireand,bymeansofmirrors,dividesitintotwoparallelbeamsofequal-intensityradiation.Thesampleisplacedinonebeam,andtheotherbeamisusedasareference.Thebeamsthenpassintothemonochromator,whichdisperseseachintoacontinuousspectrumoffrequenciesofinfraredlight.Themonochromatorconsistsofarapidlyrotatingsector(beamchopper) thatpassesthetwobeamsalternatelytoadiffractiongrating(aprisminolderinstruments).Theslowlyrotatingdiffractiongratingvariesthefrequencyorwavelengthofradiationreachingthethermocoupledetector.Thedetectorsensestheratiobetweentheintensitiesofthereferenceandsamplebeams.Inthisway,thedetectordetermineswhichfrequencieshavebeenabsorbedbythesampleandwhichfrequenciesareunaffectedbythelightpassingthroughthesample. Afterthesignalfromthedetectorisamplified,therecorderdrawstheresultingspectrumofthesampleonachart.Itisimportanttorealizethatthespectrumisrecordedasthefrequencyofinfraredradiationchangesbyrotationofthediffractiongrating.Dispersiveinstrumentsaresaidtorecordaspectruminthefrequencydomain.
- 35. •Notethatitiscustomarytoplotfrequency(wavenumber,cm−1)versuslighttransmitted,notlightabsorbed.Thisisrecordedaspercenttransmittance(%T)becausethedetectorrecordstheratiooftheintensitiesofthetwobeams,and %Transmittance=Is/Irx100 •whereIstheintensityofthesamplebeam,andIristheintensityofthereferencebeam.Inmanypartsofthespectrum,thetransmittanceisnearly100%,meaningthatthesampleisnearlytransparenttoradiationofthatfrequency(doesnotabsorbit).Maximumabsorptionisthusrepresentedbyaminimumonthechart.Evenso,theabsorptionistraditionallycalledapeak.Thechemistoftenobtainsthespectrumofacompoundbydissolvingitinasolvent. •Thesolutionisthenplacedinthesamplebeam,whilepuresolventisplacedinthereferencebeaminanidenticalcell.Theinstrumentautomatically“subtracts”thespectrumofthesolventfromthatofthesample.Theinstrumentalsocancelsouttheeffectsoftheinfrared- activeatmosphericgases,carbondioxideandwatervapor,fromthespectrumofthesample(theyarepresentinbothbeams).Thisconveniencefeatureisthereasonmostdispersiveinfraredspectrometersaredouble-beam(sample+reference)instrumentsthatmeasureintensityratios;sincethesolventabsorbsinbothbeams,itisinbothtermsoftheratioIs/Irandcancelsout.Ifapureliquidisanalyzed(nosolvent),thecompoundisplacedinthesamplebeam,andnothingisinsertedintothereferencebeam.Whenthespectrumoftheliquidisobtained,theeffectsoftheatmosphericgasesareautomaticallycanceledsincetheyarepresentinbothbeams
- 37. B. Fourier Transform Spectrometers •Themostmoderninfraredspectrometers(spectrophotometers)operateonadifferentprinciple.Thedesignoftheopticalpathwayproducesapatterncalledaninterferogram.Theinterferogramisacomplexsignal,butitswave-likepatterncontainsallthefrequenciesthatmakeuptheinfraredspectrum. •Aninterferogramisessentiallyaplotofintensityversustime(atime- domainspectrum).However,achemistismoreinterestedinaspectrumthatisaplotofintensityversusfrequency(afrequency-domainspectrum).AmathematicaloperationknownasaFouriertransform(FT) canseparatetheindividualabsorptionfrequenciesfromtheinterferogram,producingaspectrumvirtuallyidenticaltothatobtainedwithadispersivespectrometer.ThistypeofinstrumentisknownasaFouriertransforminfraredspectrometer,orFT-IR. •TheadvantageofanFT-IRinstrumentisthatitacquirestheinterferograminlessthanasecond.Itisthuspossibletocollectdozensofinterferogramsofthesamesampleandaccumulatetheminthememoryofacomputer. WhenaFouriertransformisperformedonthesumoftheaccumulatedinterferograms,aspectrumwithabettersignal-to-noiseratiocanbeplotted.AnFT-IRinstrumentisthereforecapableofgreaterspeedandgreatersensitivitythanadispersioninstrument.
- 38. •AschematicdiagramofanFT-IRisshowninFigure2.3b. •TheFT-IRusesaninterferometertoprocesstheenergysenttothesample.Intheinterferometer,thesourceenergypassesthroughabeamsplitter,amirrorplacedata45°angletotheincomingradiation,whichallowstheincomingradiationtopassthroughbutseparatesitintotwoperpendicularbeams,oneundeflected,theotherorientedata90°angle. •Onebeam,theoneorientedat90°inFigure2.3b,goestoastationaryor“fixed”mirrorandisreturnedtothebeamsplitter.Theundeflectedbeamgoestoamovingmirrorandisalsoreturnedtothebeamsplitter. •Themotionofthemirrorcausesthepathlengththatthesecondbeamtraversestovary.Whenthetwobeamsmeetatthebeamsplitter,theyrecombine,butthepathlengthdifferences(differingwavelengthcontent) ofthetwobeamscausebothconstructiveanddestructiveinterferences. •Thecombinedbeamcontainingtheseinterferencepatternsiscalledthe interferogram.Thisinterferogramcontainsalloftheradiativeenergycomingfromthesourceandhasawiderangeofwavelengths.
- 39. •Theinterferogramgeneratedbycombiningthetwobeamsisorientedtowardthesamplebythebeamsplitter.Asitpassesthroughthesample,thesamplesimultaneouslyabsorbsallofthewavelengths(frequencies)thatarenormallyfoundinitsinfraredspectrum. •Themodifiedinterferogramsignalthatreachesthedetectorcontainsinformationabouttheamountofenergythatwasabsorbedateverywavelength(frequency). •Thecomputercomparesthemodifiedinterferogramtoareferencelaserbeamtohaveastandardofcomparison.Thefinalinterferogramcontainsalloftheinformationinonetime-domainsignal,asignalthatcannotbereadbyahuman. •AmathematicalprocesscalledaFouriertransformmustbeimplementedbycomputertoextracttheindividualfrequenciesthatwereabsorbedandtoreconstructandplotwhatwerecognizeasatypicalinfraredspectrum.Computer-interfacedFT-IRinstrumentsoperateinasingle-beammode.
- 40. •Toobtainaspectrumofacompound,thechemistfirstobtainsaninterferogramofthe“background,”whichconsistsoftheinfrared- activeatmosphericgases,carbondioxideandwatervapor(oxygenandnitrogenarenotinfraredactive). •TheinterferogramissubjectedtoaFouriertransform,whichyieldsthespectrumofthebackground.Thenthechemistplacesthecompound(sample)intothebeamandobtainsthespectrumresultingfromtheFouriertransformoftheinterferogram. •Thisspectrumcontainsabsorptionbandsforboththecompoundandthebackground.Thecomputersoftwareautomaticallysubtractsthespectrumofthebackgroundfromthesamplespectrum,yieldingthespectrumofthecompoundbeinganalyzed. Thesubtractedspectrumisessentiallyidenticaltothatobtainedfromatraditionaldouble-beamdispersiveinstrument.
- 41. PREPARATION OF SAMPLES FOR INFRARED SPECTROSCOPY •Todeterminetheinfraredspectrumofacompound,onemustplacethecompoundinasampleholder,orcell.Ininfraredspectroscopy, thisimmediatelyposesaproblem. •Glassandplasticsabsorbstronglythroughouttheinfraredregionofthespectrum.Cellsmustbeconstructedofionicsubstancestypicallysodiumchlorideorpotassiumbromide. •Potassiumbromideplatesaremoreexpensivethansodiumchlorideplatesbuthavetheadvantageofusefulnessintherangeof4000to400cm−1. •Sodiumchlorideplatesareusedwidelybecauseoftheirrelativelylowcost.Thepracticalrangefortheiruseinspectroscopyextendsfrom4000to650cm−1. •Sodiumchloridebeginstoabsorbat650cm−1,andanybandswithfrequencieslessthanthisvaluewillnotbeobserved.Sincefewimportantbandsappearbelow650cm−1,sodiumchlorideplatesareinmostcommonuseforroutineinfraredspectroscopy.
- 44. Advantages of Fourier transform IR over dispersive IR •ThemultiplexorFellgett'sadvantage.Thisarisesfromthefactthatinformationfromallwavelengthsiscollectedsimultaneously.ItresultsinahigherSignal-to-noiseratioforagivenscan-timeorashorterscan-timeforagivenresolution. •ThethroughputorJacquinot'sadvantage.Thisresultsfromthefactthat, inadispersiveinstrument,themonochromatorhasentranceandexitslitswhichrestricttheamountoflightthatpassesthroughit.Theinterferometerthroughputisdeterminedonlybythediameterofthecollimatedbeamcomingfromthesource. •ConnesadvantageThewavenumberscaleofaninterferometerisderivedfromaHe-Ne(heliumneon)laserthatactsasaninternalreferenceforeachscan.Thewavenumberofthislaserisknownveryaccuratelyandisverystable.Asaresult,thewavenumbercalibrationofinterferometersismuchmoreaccurateandhasmuchbetterlongtermstabilitythanthecalibrationofdispersiveinstruments.
- 45. •NegligiblestraylightBecauseofthewayinwhichtheinterferometermodulateseachsourcewavelength.Thereisnodirectequivalentofthestraylightfoundindispersivespectrometers. •HighResolution. •Goodforweakabsorptionbands. •Canstudysamplesunderhighabsorbance. •Lesstimeforscan. •NodiscontinuitiesBecausetherearenogratingorfilterchanges,therearenodiscontinuitiesinthespectrum.
- 46. Disadvantages of FT-IR •Detectors response time needs to be fast. •Initial cost is higher. •Maintenance is complicated.
- 48. CORRELATION CHARTS AND TABLES •Toextractstructuralinformationfrominfraredspectra,youmustbefamiliarwiththefrequenciesatwhichvariousfunctionalgroupsabsorb.Youmayconsultinfraredcorrelationtables,whichprovideasmuchinformationasisknownaboutwherethevariousfunctionalgroupsabsorb.Theabsorptioninformationispresentedintheformofachartcalledacorrelationchart.
