Ewing's sarcoma
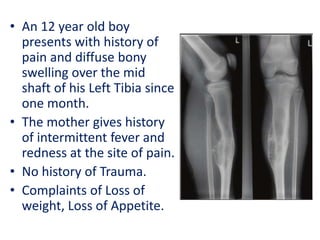
- 1. • An 12 year old boy presents with history of pain and diffuse bony swelling over the mid shaft of his Left Tibia since one month. • The mother gives history of intermittent fever and redness at the site of pain. • No history of Trauma. • Complaints of Loss of weight, Loss of Appetite.
- 2. Differential Diagnosis •Osteomyelitis •Osteoid osteoma •Eosinophilic Granuloma •Ewing’s tumour •Osteosarcoma
- 3. Osteomyelitis Osteoid osteoma Ewing’s tumour Osteosarcoma Eosinophilic Granuloma Presentation Pain, Redness, Fever, Weakness. Dull pain, Limping, swelling. Pain, Swelling, Pathological fracture. Pain, swelling, worse at night Pain Age All ages 5-25 years 8-20 years Bimodal distribution (<25 &>60 yrs) Adolesents/ Young adults. Sex Males Males(3:1) Males(1.5- 2:1) Males Equal Duration Short/Long Long Short Short short Benign/ Malignant Infective origin Commonest benign Second most Bone cancer in childern. Most common Bone cancer in childern. Benign bone lesion soild periosteal reaction Site Usually at metaphysis Diaphysis Diaphysis - Femur, tibia, flat bone Metaphysis: Femur, tibia (knee) Skull, ribs, Femur. (Metaphysis) Others No h/o trauma Pain relieved with NSAIDs Can have H/O Trauma Most with H/O Trauma H/O fever, Trauma.
- 5. • Ewing’s sarcoma is the second most common bone cancer of children, following osteosarcoma. • The median age at presentation is 14 years (usually 8–25 years) • Boys are affected more than girls (1.5–2:1)
- 6. • American Pathologist • Suffered from OM at the age of 14yrs. • Confined to bed for 2 yrs. • Served as Prof of Pathology for 33 yrs at Cornell Univ. New York. • Died of bladder cancer at 76yrs. • Described in 1921 as an undifferentiated tumor involving the diaphysis of long bones that is radiation sensitive (in contrast to osteosarcoma). James Stephen Ewing (1866-1943)
- 7. At Presentation: • Pain (>90%) • Swelling or mass (65%) • Limitation in movement (25%) • Neurologic changes (15% overall, though 50% in central tumors) • Pathologic fracture (15%) • Fever (10%) • Approximately 25% have overt metastases at presentation.
- 10. • Usually show an area of bone destruction which is predominantly in the mid-diaphysis. • New bone formation may extend along the shaft and sometimes appears as fusiform layers of bone around the lesion – ‘onion-peel’ effect. • Often the tumour extends into the surrounding tissues, with radiating streaks of ossification (‘sunray’ appearance) and reactive periosteal bone at the proximal and distal margins (Codman’s triangle).
- 12. The definitive diagnosis is based on • Histomorphologic findings • Immunohistochemistry • Molecular pathology HISTOMORPHOLOGICAL FINDINGS: • Neuroectodermal tissue is the embryonic tissue of origin for EWS and it is derived from primordial BM mesenchymal stem cells. • Ewing's sarcoma is a small-blue-round-cell tumors • Have a clear cytoplasm on H&E staining, due to glycogen. • The presence of the glycogen can be demonstrated with • Positive PAS staining and • Negative PAS diastase staining. • Differential for small round blue cell tumors (mnemonic LEMONS): Lymphoma, Ewing’s, Medulloblastoma, Other (rhabdomyosarcoma, pineoblastoma, ependymoblastoma, etc.), Neuroblastoma, Small cell carcinoma.
- 16. • The characteristic immunostain is CD99, which diffusely marks the cell membrane. (CD99 is not specific for Ewing's sarcoma) • The most common translocation, present in about 90% of Ewing sarcoma cases, is t(11;22)(q24;q12) • Generates an aberrant transcription factor through fusion of the EWSR1 gene with the FLI1 gene.[FLI1(11): EWS(22)] • Other minor translocations include: • t(21;22), c-Myc proto-oncogene(chromosome 21) and EWS gene(chromosome 22) in 10% of cases. • t(7;22)
- 17. • Ewing’s family of tumors includes • Ewing’s sarcoma (bone 87%), • Extraosseous Ewing’s sarcoma (8%), • Peripheral PNET (5%), and • Askin’s tumor (PNET of chest wall). • Markers that differentiate EWS: 1. Vimentin 2. HBA-71 3. β2-microglobulin 4. ↑c-myc (vs. n-myc in NB) • PNET and EWS have similar translocations and are both CD99 (MIC2)+ and vimentin+. • PNET is NSE+, S100+, more differentiated, and has more neuroendocrine features. • EWS is NSE−, S100 variable, and Homer Wright rosettes+.
- 18. At presentation • 75-80% of patients present with localized disease. • 25% of EWS pts present with mets. Lung (25%–40%) ≥ Bone/BM (∼25%) and LNs (<10%). • Bone Marrow micromets are seen in 25% (localized) & 40% (lung mets) 5-yr OS: • Localised disease is 60-70% • Metastatic EWS: 1. Lung mets: ∼35% 2. Bone/BM mets: ∼25% 3. Lung + bone/BM mets: ∼15% (Paulussen M et al., Ann Oncol 2009)
- 19. Poor prognosis in EWS • Male gender • Age >15 yrs (>17 yrs in some) • Pelvic/axial Site or rib origin • Size (>8 cm per St. Jude or >100 cc per CESS- 81 [Cooperative Ewing Sarcoma Studies]) • Stage (presence/absence of metastatic Dz is strongest prognostic factor) • ↑LDH • Poor response to chemo (>10% viable tumor) • No surgery
- 21. Chemotherapy • IESS-1 (JCO 1990): – Nonrandomized comparison of 342 patients with localized disease treated with VAC + D vs. VAC vs. VAC + prophylactic bilateral whole lung RT plus noncontrolled local intervention. – 5-year RFS was best with VAC + D (60%) vs. VAC (24%) vs. VAC + RT (44%). – VACA is superior to VAC or VAC + BPR. VAC + bilateral pulmonary RT is superior to VAC. No improvement between treatment modalities was noted for pelvic cases. • IESS-2 (JCO 1991): – 214 patients with localized nonpelvic primaries randomized to high-dose, intermittent VAC + D vs. moderate dose continuous VAC + D. – Local treatment was surgery ± postoperative RT or RT alone (whole bone to 45 Gy with primary boost to 55 Gy). – High-dose VAC + D improved OS (63 → 77%); no difference in OS for local control modalities. – High dose intermittent VACA is superior to the IESS-I regimen, even for pelvic tumors. OS actually superior to nonpelvic pts in IESS-I. – Take home: Intermittent VACA is standard of care. • IESS-3/INT 0091 (NEJM 2003): – 518 patients with localized or metastatic disease randomized to VDCA vs. VDCA alternating with IE. – Local treatment was given at week 9–15 with RT, surgery, or both. – Adding IE improved 5-year OS (61 → 72%) for localized disease, but not for metastatic disease (25%). – VACA+IE superior to VACA for OS and localized disease, but no benefit in metastatic disease. – Take home: high dose, intermittent VACA+IE is standard of care.
- 22. • IESS-IV tested dose intensification of VAdriaC+IE • AEWS0031 tested interval compression of VAdriaC+IE These showed that VAdriaC+IE q2wks was superior and forms the current standard of care in the definitive setting. • COG protocol AEWS0031 a randomized trial for patients with newly diagnosed localized Ewing sarcoma. • This trial sought to intensify therapy not by dose escalation, but by decreasing the interval between chemotherapy cycles (interval compression). • Patients randomized to receive VDC/IE cycles alternating every 3 weeks vs 2 weeks. • 5-year event-free survival (73% versus 65% for patients randomized to the standard arm). • This trial established interval compressed VDC/IE as a new standard approach for patients with localized Ewing sarcoma.
- 23. • VAC/IE (vincristine + doxorubicin [Adriamycin] + cyclophosphamide alternating with ifosfamide + etoposide) • Alternating VAC and IE cycles. • Repeat each cycle every 3 weeks for 17 cycles. VAC cycles • Day 1: Vincristine 2mg/m2 (max 2mg) IV + doxorubicin 75mg/m2 IV bolus + cyclophosphamide 1,200mg/m2 IV. – Dactinomycin can be substituted for doxorubicin if there are concerns regarding cardiotoxicity – Dactinomycin 1.25mg/m2 IV can be substituted for doxorubicin when a total doxorubicin dose of 375mg/m2 is reached. IE cycles • Days 1–5: Ifosfamide 1,800mg/m2 IV + mesna + etoposide 100mg/m2 IV.
- 25. Surgery: • For local control, Surgery preferred unless poor functional results are anticipated. • Resection provides pathologic information post-chemotherapy, avoids second malignancy and late effects of RT. • Resection without reconstruction can be done in small bones such as rib, clavicle, proximal fibula, distal scapula, metatarsals, metacarpals, and small iliac wing or pubic bone lesions. • Results are typically very good for these “dispensable bones.” • Large lesions may require allograft or endoprosthetic reconstructions. • Metastatic setting: – Limited pulmonary metastases – Palliation at primary site • Nodal dissection is not routinely indicated. LOCAL THERAPY
- 26. • Adequate surgical margins in EWS Per COG protocol AEWS0031(Womer RB et al., JCO 2012) – >1 cm for bone – >0.5 cm for Sdft Tissue – >0.20 cm for fascia • Sg is preferred – Expendable bones are involved – If there is a pathologic fracture • 3 indications for adj RT after Sg in EWS: – +Margin – Tumor spill – >10% viable tumor after induction chemo (poor chemo response).
- 27. Radiation: • RT potentially indicated pre-op, post-op, or definitively for the primary tumor and for treatment of pulmonary and skeletal metastases. • Indications for postoperative RT – Close margins (<1 cm) – Poor histologic response (<90% necrosis) – Tumor spill. • Preoperative RT considered when close/positive margins are expected. • Treat pre-chemo volume due to high rate of local failure if limited to post-chemo volume. • Involved field rather than whole bone is sufficient. • Hyperfractionation does not improve outcomes, but may improve fracture rate, range of motion, and muscle atrophy. • Adjuvant RT starts at the time of consolidation CHT (week 14). • VC-IE CHT is given during RT (doxorubicin held during RT).
- 28. No randomized trials have directly compared RT to surgery for LC of Ewing’s sarcoma. • CESS 86 (JCO 2001): – 177 patients with localized Ewing’s treated with chemotherapy – Nonrandomized local control arms of • Surgery alone • Surgery plus 45 Gy RT • Definitive RT of 60 Gy (randomized to qd vs. b.i.d.). – RT used 5 cm proximal/distal margins and 2 cm lateral/deep margins. – 5-year OS was 69% with no differences in OS or RFS according to local therapy. – Local control was 100% for surgery, 95% for surgery plus RT, and 86% for RT alone (no difference for qd vs. b.i.d. RT).
- 29. • POG-8346 (Donaldson SS et al., IJROBP 1998) – Osseous EWS pts after induction chemo rcvd definitive RT for local therapy randomized to • Whole bone RT (39.6 Gy → 55.8 Gy boost to GTV +2 cm) vs. • Involved-field RT (GTV + 2 cm to 55.8 Gy) • All pts then rcvd maintenance chemo. • The RT Tx techniques had similar 5-yr EFS (∼41%) and LC (∼53%).
- 30. CESS-81 and -86 and EICESS 92 (European Intergroup CESS) • Schuck et al. (IJROBP 2003) • Performed a secondary analysis of 1,085 pts • Compared PreOP RT vs. PORT vs. Definitive RT • Local Failure – Similar b/t preop and postop RT (5.3% vs. 7.5%) but – Significantly worse in the definitive RT arm (26%) • Criticism: There was a strong negative selection bias against the definitive RT cohort. • There was no difference in LF b/t RT alone and Sg + post-RT if only partial resection was achieved. • In CESS-81, Disease free survival after 41 months for patients with – Tumor volume <100 ml was 75% – Tumor volume greater than or equal to 100 ml was 10% • Preop RT may improve LC if unresectable.
- 31. • Talleur (IJROBP 2016): –St. Judes phase II trial of 45 EWS patients –Randomised to 55.8 vs. 64.8 Gy based on tumor size <8 vs. ≥8 cm, respectively. –All patients treated with 1 cm margins on gross tumor. –LF rate 4.4% at 10 year.
- 32. Chest wall Ewings Sarcoma (Rib primary & Askin’s tumor) • More commonly seen in females. • Preoperative CHT – Improves negative margins (50% vs. 77%) – Decreased need for post-op RT (5-yr EFS 56%) • Schuck A et al. retrospectively reviewed 138 pts with localized CW EWS treated in CESS-86 and EICESS 92. (IJROBP 2002) – 42 pts rcv hemithorax RT. – If <14 yo, then 15 Gy; otherwise, 20 Gy at 1.5 Gy/fx or 1.25 Gy bid. – All RT pts rcv a boost to the primary site of 45–60 Gy. – 7-yr EFS trended in its favor (63% vs. 46%). – Improvements in EFS appeared to be d/t reductions in pulmonary mets. – A major criticism of this study is that the RT group had sup chemo.
- 33. METASTATIC DISEASE • If patient has Lung Mets: • Chemotherapy → WLI or surgical resection (if <5 mets). • EICESS secondary analyses: Paulussen et al. (a) isolated pulmonary mets or (b) combined lung + bone/BM mets – Treated +/– WLI as part of a series of protocols from the EICESS. – WLI was associated with improved EFS in both subgroups. (Ann Oncol 1998) • St. Jude’s retrospective study: Rodriguez-Galindo et al. – EWS pts with isolated pulmonary recurrence. – Pts who rcvd WLI had improved 5-yr postrecurrence survival (30% vs. 17%). (Cancer 2002)
- 34. • The WLI dose in EWS depends on age: – If <14 yo, then 15 Gy (1.5 Gy/fx) – If ≥14 yo, then 18 Gy (mostly in European protocols, but US protocols still use 15 Gy). The field borders used in WLI for EWS. • Superior–Inferior: 1 cm above 1st rib to L2 (Block PA kidney at 7.5 Gy) • Lateral: 1 cm lat rib cage.
- 35. RADIATION DOSES • Radiation fields customized depending on primary site • MRI is recommended for treatment planning in all cases • Definitive RT for bone tumors with no soft-tissue involvement: – Prechemotherapy GTV plus a 2 cm margin to 55.8 Gy. • Definitive RT for bone tumors with a soft-tissue component: – Prechemotherapy GTV plus a 2 cm margin to 45 Gy boost to 55.8 Gy to the initial bony GTV + postchemotherapy soft-tissue extent with a minimal margin. • For postoperative RT – Pretreatment GTV plus a 2 cm margin to 45 Gy boost -Postoperative microscopic/tumor spill + 2 cm margin to 50.4 Gy -Gross residual disease + 2 cm margin to 55.8 Gy. • 45 Gy for vertebral body involvement b/c of SC tolerance.
- 36. • LN positive—resected: Nodal Bed-50.4 Gy in 1.8 Gy/fx • LN positive—unresected: Nodal level: 45 Gy, Gross node: 55.8 Gy in 1.8 Gy/fx • Malignant ascites/diffuse peritoneal involvement: – Whole abdomen RT: 25 Gy in 1.5 Gy/ fx • Lung metastases: Whole lung RT: 15 Gy in 1.5 Gy/fx – (if <6 years, then 12 Gy in 1.5 Gy/fx) • Chest wall tumor/pleural nodules/effusion: – Hemithorax RT: 15 Gy in 1.5 Gy/fx – Chest wall tumor: • Prechemo volume +1.5-2cm=30.6 Gy • Postchemo Volume+1.5-2cm=36 Gy in 1.8 Gy/fx – Pleural nodule: • Prechemo volume +1.5-2cm=21.6 Gy • Postchemo Volume+1.5-2cm=36 Gy in 1.8 Gy/fx. AEWS1031 contouring guidelines
- 37. COMPLICATIONS 1) Dermatitis and recall reaction: ADR and dactinomycin A. 2) Cystitis: Cyclophosphamide or ifosfamide or RT cystitis. 3) Abnormal bone and soft-tissue growth and development. – Premature epiphyseal closure: occurs at >20 Gy. – Decreased bone growth can occur at ∼10 Gy. – Limb length discrepancy of 2–6 cm can be managed with a shoe lift. – Consider amputation and prosthesis in the very young as they recover function well.
- 38. 4) Permanent weakening of affected bone. – The highest risk for fracture is within 18 months of RT, during which time patients should avoid contact and high-impact sports. – Factors that influence fracture risk • Total dose, extent of cortical disruption at Dx, younger age, and 2nd bone malignancy in the RT field. 5) Decreased range of motion secondary to soft-tissue and/or joint fibrosis. 6) Lymphedema can be prevented by – Sparing a 1–2-cm strip of skin on the extremity or – Minimize the circumferential RT dose to 20–30 Gy
- 39. 7) Second malignancy • Kuttesch et al. retrospectively reviewed 266 EWS pts treated at St. Jude’s Hospital. • 20-yr cumulative incidence of 2nd malignancies was – 9.2% for any malignancy and – 6.5% for sarcoma • RT dose–response relationship with a 2nd malignancy RR of 40 if RT was >60 Gy and minimal for <48 Gy. (JCO 1996) • Most common second tumor is osteosarcoma.
- 40. Chemotherapy Side effects: – Vincristine (neuropathy, constipation, myalgias, arthralgias, and cholestasis) – Cyclophosphamide (pancytopenia and dose-dependent hemorrhagic cystitis, infertility) – Doxorubicin (myocardial dysfunction and pancytopenia) – Ifosfamide (high incidence of hemorrhagic cystitis requiring use of Mesna and Fanconi syndrome of electrolyte wasting) – Etoposide (pancytopenia, anaphylactic reactions, and second malignancies such as AML).
- 41. FOLLOW-UP • H&P + CXR every 3 months for 2 years. • X-ray primary every 3 months (and/or MRI every 6 months) for 2 years. • After 2 years, may increase follow-up intervals, but should obtain CBC annually.
Hinweis der Redaktion
- Ewing’s sarcoma commonly presents in Lower extremity (femur 15–20%, tibia or fibula 5–10%), Pelvis (20–30%), Upper extremity (humerus 5–10%), Ribs (9–13%), and Spine (6–8%).
- S100 calcium binding protein B Neuron-Specific Enolase
- 75-80% of patients present with localized disease, but 20–25% have gross metastases to the lung, bone, or bone marrow, and nearly all patients have micrometastases at diagnosis, so all patients require chemotherapy.
- dose intensity defined as the amount of drug delivered per unit of time [mg/m²/week].
- Local Failure rate for EWS after definitive RT: 5%–25% Worse with pelvic sites (LF 15%–70%) Worse with large (>8 cm) lesions (LF 20%).
- Cooperative Ewing Sarcoma Study
- In a recent review of RT-induced osteosarcoma, most common primary was Ewing’s (25%), median latency was 8 years. 5-yr OS was 40% overall, with aggressive CHT and surgery 5-yr OS 68% versus chemo alone 17%.
