Jak stat
- 1. 1
- 2. ProteinTyrosine Kinase, of which there are about 90 in human genome, phosphorylate specific tyrosine residues on target proteins that can then activate one or more signalling pathways. There are two large and important classes of cell-surface-receptors that activate protein tyrosine kinase are: 1. ReceptorTyrosine kinases (RTKs) 2. Cytokine receptor (JAK Kinase) 2
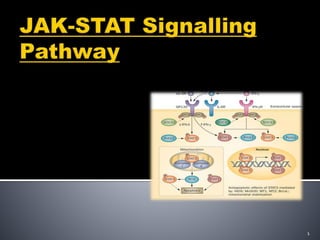
- 3. The JAK-STAT(Janus kinase–signal transducer and activator of transcription) signalling pathway transmits information from chemical signals outside the cell, which causes DNA transcription and activity in the cell. The JAK-STAT system is a major signalling alternative to the second messenger system. 3
- 4. (1) a receptor (2) Janus kinase (JAK) and (3) Signal Transducer and Activator of Transcription (STAT). 4
- 5. JAKs, which have tyrosine kinase activity, bind to some cell surface cytokine receptors.The binding of the ligand to the receptor triggers activation of JAKs. With increased kinase activity, they phosphorylate tyrosine residues on the receptor STATs possessing SH2 domains are recruited to the receptors, and are themselves tyrosine-phosphorylated by JAKs. Activated STAT dimers accumulate in the cell nucleus and activate transcription of their target genes. STATs may also be tyrosine-phosphorylated directly by receptor tyrosine kinases, such as the epidermal growth factor receptor, as well as by non-receptor tyrosine kinases. 5
- 6. 6
- 7. 7
- 8. The receptor is activated by a signal from interferon, interleukin, growth factors, or other chemical messengers. 8
- 9. 9 Regulatory molecule that bind to receptors linked to JAK.
- 10. 10
- 11. The pathway is negatively regulated on multiple levels. Protein tyrosine phosphatases remove phosphates from cytokine receptors and activated STATs. SHP1, a protein tyrosine phosphatase, is present in an inactive form in unstimulated cells. Binding of an SH2 domain in SHP1 to a particular phosphotyrosine in the activated receptor unmasks its phosphatase catalytic site . Removal of the phosphate from this tyrosine inactivates the JAK kinase. SOCS proteins, whose expression is induced in erythropoietin-stimulated erythroid cells, inhibit or permanently terminate signaling over longer time periods. Binding of SOCS to phosphotyrosine residues blocks binding of other signaling proteins. 11
- 12. 12
- 13. In mammals, there are seven STAT genes, and each one binds to a different DNA sequence.This affects basic cell functions, like cell growth, differentiation and death. In mammals, the JAK/STAT pathway is the principal signaling mechanism for a wide array of cytokines and growth factors. JAK activation stimulates cell proliferation, differentiation, cell migration and apoptosis. Mutations that constitutively activate or fail to regulate JAK signaling properly cause inflammatory disease, erythrocytosis, gigantism and an array of leukemias. The JAK-STAT pathway is evolutionarily conserved, from slime molds and worms to mammals (but not fungi or plants). Disrupted or dysregulated JAK-STAT functionality (which is usually by inherited or acquired genetic defects) can result in immune deficiency syndromes and cancers. 13
- 14. Ras is a monomeric, GTP-binding switch protein. Ras alternates between an inactive state with bound GDP and an active state with bound GTP. Ras is not directly linked to cell-surface receptors. Ras is anchored to the plasma membrane by a hydrophobic anchor. 14
- 15. Ras acts as a binary signal switch cycling between ON and OFF states, which are characterized in terms of a small molecule, a guanine nucleotide, bound to the protein. In the resting cell, Ras is tightly bound to GDP which is exchanged with GTP upon binding of extracellular stimuli to cell membrane receptors. In the GTP-bound form, Ras interacts specifically with so-called effector proteins, thereby initiating cascades of protein-protein interactions that may finally lead to cell proliferation. To return to the inactive OFF state, Ras cleaves off the terminal phosphate moeity, the intrinsic GTPase reaction.The remaining GDP- bound Ras is no longer able to interact with effectors, it is switched OFF. 15
- 16. 16
- 17. MAP kinase pathway is activated: Ras activates Raf protein, a serine/ threonine kinase, by binding to it. Hydrolysis of RasGTP causes the release of active Raf. Raf activates MEK, another kinase. MEK activates MAP Kinase (MAPK). MAPK translocates to the nucleus , causing induction of gene transcription.
- 18. 18
- 19. 19 The JAK-STATSignaling Pathway: Input and Output Integration The JAK-STAT Signaling Pathway: Input Peter J. Murray http://www.jimmunol.org/content/178/5/2623 doi: 10.4049/jimmunol.178.5.2623 The JAK/STAT signaling pathway Jason S. Rawlings, Kristin M. Rosler and Douglas A. Harrison* University of Kentucky, Department of Biology, 101 T.H. Morgan Bldg., Lexington, KY 40506, USA Journal of Cell Science 117, 1281-1283 Published by The Company of Biologists 2004 doi:10.1242/jcs.00963 Molecular Cell Research Volume 1773, 1177- 1193, Ras Oncogenes and their downstream Krishnaraj Rajalingam, Ralf Schreck, Ulf R. Rapp Stefan Albert. Molecular Cell Biology By Lodish et. al.
- 20. 20
Hinweis der Redaktion
- It acts through the cell membrane, and into gene promoters on the DNA in the cell nucleus.
- Many JAK-STAT pathways are expressed in white blood cells, and are therefore involved in regulation of the immune system.
- and create sites for interaction with proteins that contain phosphotyrosine-binding SH2 domains. capable of binding these phosphotyrosine residues These phosphotyrosines then act as binding sites for SH2 domains of other STATs, mediating their dimerization. Different STATs form hetero- or homodimers.
- This activates the kinase function of JAK, which autophosphorylates itself (phosphate groups act as "on" and "off" switches on proteins). The STAT protein then binds to the phosphorylated receptor, where STAT is phosphorylated by JAK. The phosphorylated STAT protein binds to another phosphorylated STAT protein (dimerizes) and translocates into the cell nucleus. In the nucleus, it binds to DNA and promotes transcription of genes responsive to STAT.
- More recently identified by binding and inhibiting JAKs or competing with STATs for phosphotyrosine binding sites on cytokine receptors. STATs are also negatively regulated by
- . STAT binds to a DNA sequence called a promoter, which controls the expression of other DNA sequences Predictably, mutations that reduce JAK/STAT pathway activity affect these processes (reviewed by Igaz et al., 2001; O'Shea et al., 2002). Conversely, Here we present a general overview of the JAK/STAT pathway and illustrate the primary mechanisms of activation and regulation of this essential signaling cascade. Here we present a general overview of the JAK/STAT pathway and illustrate the primary mechanisms of activation and regulation of this essential signaling cascade.